
In modern days everybody is moving towards the gadget to make their lifestyle easy and smooth. But the major challenge toward this is energy. Every gadget or accessory from smartphones to satellites needs power for its operation and is being achieved by using fossils fuel or batteries. But all of us know that we are rapidly consuming fossil fuels in comparison with their restoration rates and if we are looking towards batteries then they take a long charging time. In upcoming years we will need a power source that took less charging time with longer backup time. For this purpose, “Super-Capacitors” will become a good choice. You may be known about Capacitors.
Storing Charge:
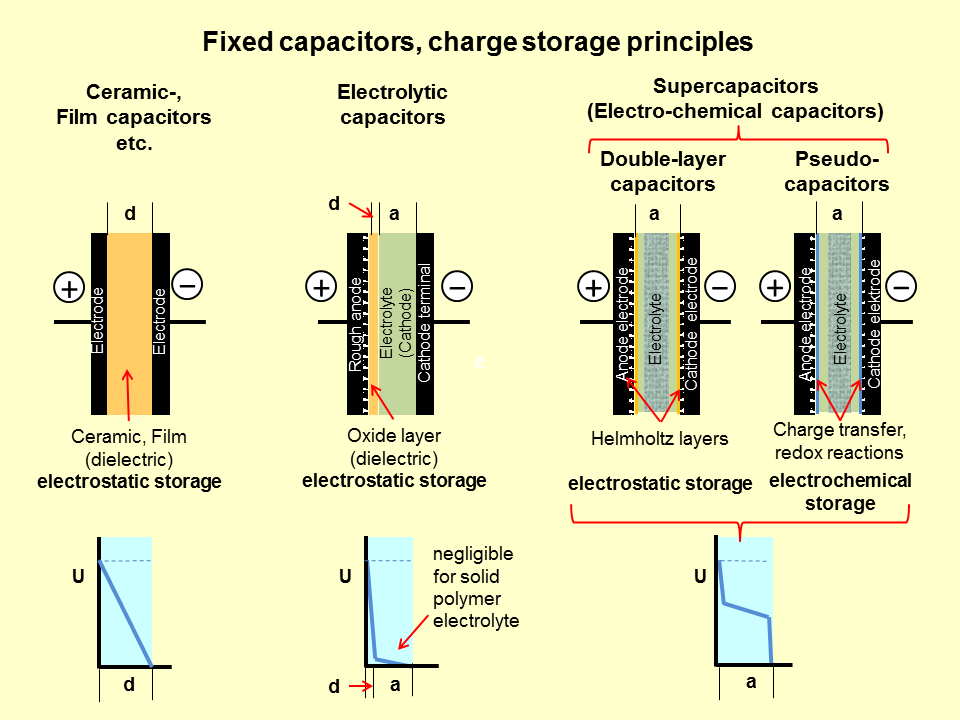
Batteries and capacitors both of them are being used to store electric charges but their storing mechanism is completely different from each other.
Batteries have two electrical terminals (Positive and Negative) separated by chemical materials known as electrolytes. When you switch on the circuit of the battery then stored chemical energy inside the battery gets converted into electrical energy. Once the chemicals have all been depleted, the inside reactions stop and the battery is discharged. In a rechargeable battery, such as Lithium-ion, the inside reactions can happily run in either direction. So you can usually charge and discharge hundreds of times before replacing it.
Capacitors are a little bit different from batteries. They use static electricity to store energy. Inside a simple electrolytic capacitor, there are two conducting plates with insulating material between them. In the simple view, you can say that a dielectric or insulating material has been placed between two metallic plates. When potential or voltage has been applied across these two metallic plates then positive and negative charges build up across the dielectric material. This material allows a capacitor of a certain size to store more charges at the same voltage, so that dielectric material makes the capacitor more efficient as a charge-storing gadget like a battery.
Super-capacitors are quite different from ordinary or simple electrolytic capacitors. How? Let’s know about this.
What is a Super-Capacitor?
A super-capacitor is also known as an ultra-capacitor or double-layer electrolytic capacitor. In an ordinary capacitor charging time is low, Weight and cost are also low but the main drawback is that it cannot provides continuous energy like a battery. This lacuna is being filled by super-capacitors that can provide continuous power like a battery. That makes a difference and it is triumph over the batteries. So, how does it happen? To know about this see its construction that is different from ordinary capacitors.
Internal structure of super-capacitor
Construction:
It is different from a simple electrolytic capacitor in two terms. The first one is that its conducting plates have a much larger area and the second one is the distance between them is much smaller, because of a special kind of dielectric material between the plates.
These plates are made from metal coated with a porous substance such as a fine granule of activated charcoal, which effectively provides much more area for storing the charges. For better understanding, suppose that a simple capacitor is like a thin cloth and electricity is water. The thin cloth can soak a small amount of water whereas a super-capacitor is like a sponge that soaks a large amount of water. Here porous plates are sponges that soak huge amounts of electric charges.
Working:
In a super-capacitor, both plates are soaked in an electrolyte and separated by a very thin insulator (made of carbon, paper, or polymers). When the plates are charged up, an opposite charge forms on either side of the separator that is known as a double-layer electric charge. This is why super-capacitors are referred to as double-layer capacitors.
When opposite charges are formed on both sides of the separator, an electric layer is formed and the plates are charged. Hence the super-capacitors are charged and have higher capacitance. These capacitors are used to provide high power and enable high load currents with low resistance that makes them a useful power source. The final cost of the super-capacitor is high because of its high charging and discharging capacitance.
Application:
In upcoming years super-capacitors can take place of batteries in the below applications:
- In industrial electronic applications
- Hybrid and electric vehicles
- Can regulate voltage in the energy grid
- Used as a power backup
Thanks for reading. See you soon with another exploration!





A marvellous weblog that helped a lot to understand about supercapacitor. Electrolytic capacitors can be used in a broad range of applications because they bridge the gap between batteries and capacitors. The future of these ultracapacitors is very promising. Keep sharing such an informative piece of knowledge.